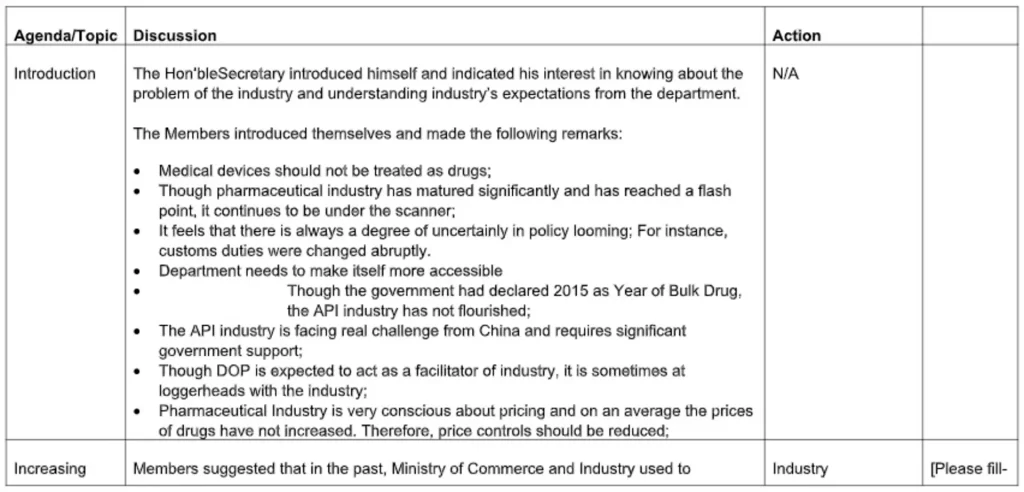
Summary
Meeting Title: LSW Round Table with K L Sharma, Joint Secretary, Ministry of Health, Dr. G N Singh, DCGI and A K Pradhan, DDCI, May 30, 2015, Mumbai
Meeting Called by: LSW LifeScienceWorld – Supported by OPPI
Meeting Date: May 30, 2015.Venue: The Orchid, Mumbai
Chair: K L Sharma, Dr. G N Singh, A K Pradhan,
Overview: Prof (Dr.) Ranjit Roy Chaudhury
Presenters:
- Dr. Suresh Menon and Yasmin Shenoy on behalf of industry (OPPI)
- Dr. S D Banavali on behalf of Investigators and Institution
Moderator:
Dr.Milind Antani on behalf of LSW LifeScienceWorld
Attendees:
Representatives from OPPI, ISCR, Sanofi, Takeda, Abbott, Wockhardt, BMS, Bayer, GSK, Pfizer, Lambda Therapeutics, Veeda Clincal Research, ACRO India and Individual investigators and Ethics Committee members.
Topics for Discussion: Issues and Recommendations on the following topics:
- Consideration of local CT data waiver for life-threatening diseases
- Training Drug Inspectors
- Inspection Checklist to be made which can be referred by the SEC committee examining the CT design
- Consideration of local CT data waiver for life-threatening diseases
- Challenges in Review and Approval Process for New Drugs/Clinical Trials (including SEC functioning)
- Accreditation of Investigators/ Sites / Ethics Committees
- Recommendations for A-V Recording
- Three Trials per Investigator
- Requirement of 50%-20% Government Sites & 50 Bedded Hospitals
- CT waiver for additional strengths / additional indications
- Post approval commitments
- GEAC working
- Company Name/Mfg. Site Name/Address Change
- Import Registration: One manufacturing Site
Action to be performed:
CDSCO is recruiting 150 new inspectors. They are ready to give one week training slot to be conducted by the industry for them.
Checklist needs to be prepared by representative from the Industry and DCGI office.
Definition of life threatening needs to be build along with a illustrative list of medical conditions.
Detailed Scope for SEC functioning needs to be spelled out. Also an orientation exercise needs to be done for all ethics committee members.
All agreed that a detailed Standard Operating procedure needs to be created for Presubmission meetings.
CDSCO to share the rationale of why only 3 trial per investigator suggested. Nishith Desai and Associates to submit a white paper on this highlighting the pain points faced by the industry.
There is already a notification made for this issue which is currently being vetted by the law ministry. The notification tries to bring clarity on the pain points being raised by the industry and academia related to consent process.
Since the DCGI approval and GEAC approval was to happen in parallel within 45 days, the inaction being practiced by GEAC is hurting the industry badly Rule change needed from 3 months import to 6 months import Current ongoing studies will not be impacted by this. There will be a window provided for completing all old approved clinical trials as per the old laws.
(Recommendation draft is being prepared by OPPI on behalf of the industry and LSW LifeScienceWorld to be sent to the Joint Secretary and DCGI office).
Participants at LSW Round Table Edition 2
- K L Sharma, Joint Secretary, MoHFW
- Dr. G N Singh, DCGI
- A K Pradhan, Asst. Drug Controller
- Dr. Ranjit Roy Chaudhury
- Dr. Milind Antani, (Moderator)
- Dr. Mubarak Naqvi, Director Medical, Sanofi
- Yasmin Shenoy, Sr. Director-Regulatory, Sanofi
- Dr. Arun Mishra, Director Regulatory, Abbott
- Dr. Rashmi Hegde, Director Medical Abbott
- Jitendra Tyagi, Managing Director, Bristol Myers-Squibb
- Ram Sundaram, Director Regulatory, BMS
- Dr. Shubnum Singh, Chief Executive, Max Healthcare
- Dr. Bikas Medhi, PGIMER, Chandigarh
- Dr. Vivek Ahuja, PATH
- Saurabh Sobti, Clinton Health Access Initiative Inc
- Dr. Jeroze Dalal, Head-Clinical Operations, GSK
- Dr. Suresh Menon, CSO, Novartis
- Dr. Ajay Sharma, Director Research, OPPI
- Rajeev Shukla, OPPI
- Dr Saurendra Das, Excel LifeSciences
- Dr. Kiran Marthak, Director- Global Clinical Development, Lambda Therapeutics
- Apurva Shah, Group Managing Director, Veeda Clinical Research
- Dr. Jyoti Bajpai, Asst. Prof. Medical Oncology, Tata Memorial Hospital
- Swapnali Raut, Director regulatory, Pfizer
- Dr. Rajen Shah, Direct-Raptim Research, President, ACRO Mumbai Chapter
- Suneela Thatte, President, ISCR
- Dr. Santosh Jha, Director, Medical, Takeda
- Dr. Vikram Singh, Vice President-Medical Affairs & Pharmacovigilance, Johnson & Johnson
- Dr. Akhilesh D Sharma, SVP & Chief medical Officer, Dr. Reddy’s
- Dr. Swashraya Shah – Executive Director, Medical Affairs, MSD
- Anirban Roychowdhury – Executive Director, Clinical Research, MSD
- Rahul Luthra – Associate Director, Regulatory Affairs, MSD
- Dr. Arani Chatterjee, Sr. Vice president, E Biologics
- Dr. Bhimsen Silgiri, Head-Medical Regulatory Affairs, Wockhardt
- Dr. Gururaj Rao, Managing Director, International Stem Cell Services Ltd
- Jayanthi Swaminathan, Apollo Hospital
- Dr. Ashish Gawde, County Medical Director, Bayer Pharmaceuticals
- Dr. Nilima Kshirsagar, National Chair Clinical Pharmacology,member DTAB
- ICMR Govt. of India, NIRRH
- Dr. B M Gandhi, Former Advisor, Dept. of Biotechnology, Govt. of India
- Dr. Vrishali Desai, Medical Director, GSK
- Dr. S D Banavali, Head – Medical Oncology, Tata Memorial Hospital
- Dr. Veena Jaguste, Director-Development Operations, Amgen
- Amita Bhave, Director Regulatory, Novartis
- Khushboo Baxi, Nishith Desai Associates
- Anay Shukla, Nishith Desai Associates
- Dr. Sonali Dighe, Medical Director, Pfizer
- Shriram Balasubramanian, Director-Commercial, Zuventus healthcare